
The hydrogen atom consists of a single negatively charged electron that moves about a positively charged proton (Figure 8.2.1 ). The available energy of excitation is somewhat less in the second case. The hydrogen atom is the simplest atom in nature and, therefore, a good starting point to study atoms and atomic structure. This page introduces the atomic hydrogen emission spectrum, showing how it arises from electron movements between energy levels within the atom. 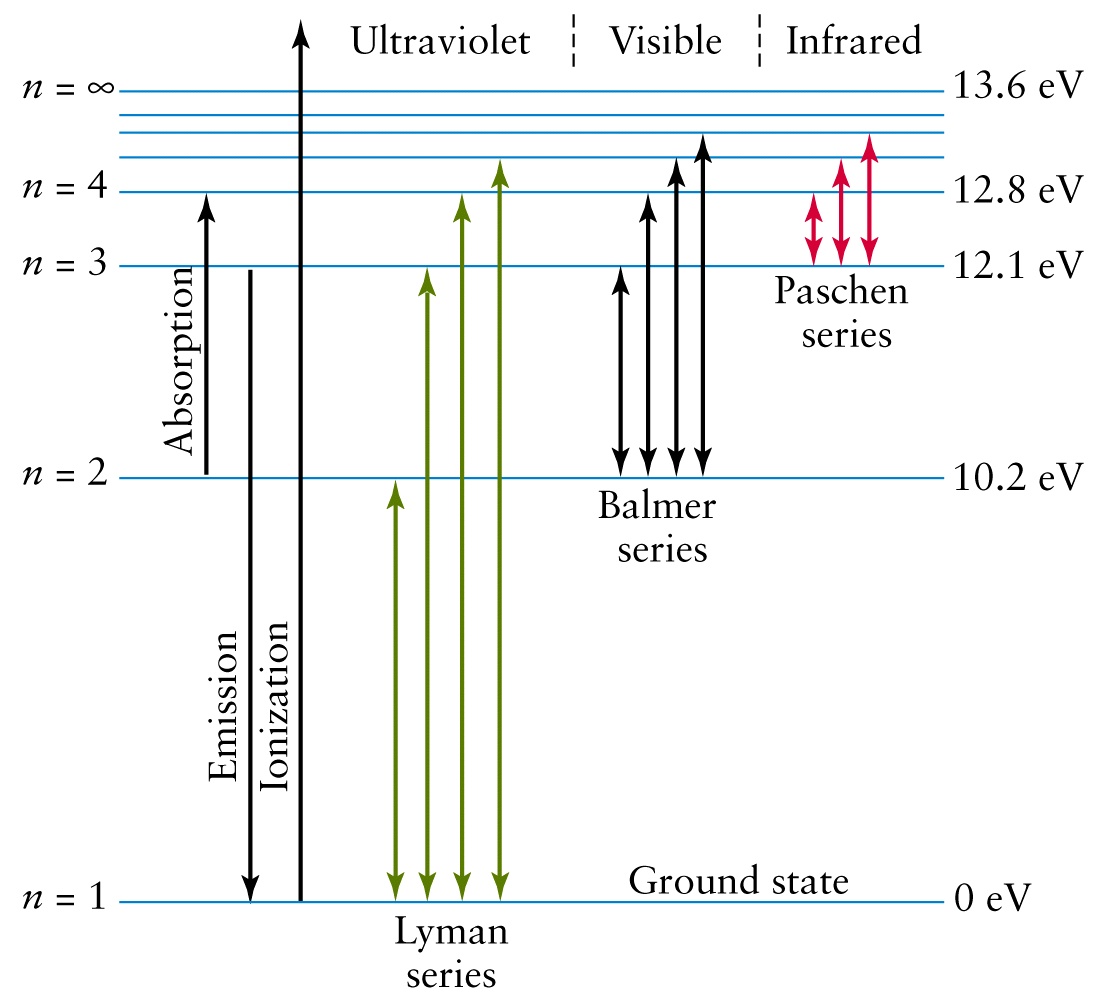
A metal hydride may first be formed and this in a two body collision with a hydrogen atom may react to give H 2 and an excited metal atom. A three body collision of two hydrogen atoms and a metal atom may result in recombination of the hydrogen and excitation of the metal atom. The observed spectral lines in the hydrogen emission spectrum are due to the atomic transitions between different energy levels. Theory of excitation of spectra by atomic hydrogen.-Two possibilities are suggested. Spectral Series of Hydrogen Atom From the image above, it is evident that the atomic hydrogen emission spectrum is divided into a number of spectral lines with wavelengths given by the Rydberg formula. Caesium, magnesium, thallium and zinc gave no line spectra. Mercury gave the complete hydride spectrum and faint emission of λ 2537. Potassium showed the first resonance doublet faintly. Sodium and cadmium gave strong emission of their first resonance lines and no other lines. Spectroscopic excitation of Na, K, Cs, Mg, Cd, Zn, Hg, Tl vapors by atomic hydrogen.-Hydrogen from a Wood discharge tube flowed into a tube containing metal vapor and the spectrum emitted by the mixture was observed. (c) doc b Bohrs model of hydrogen assumes that electrons can only exist in certain specific electronic energy levels. The Spectrum of Atomic Hydrogen For almost a century light emitted by the simplest of atoms has been the chief experimental basis for theories of the structure of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
